[fusion_builder_container hundred_percent=”no” hundred_percent_height=”no” hundred_percent_height_scroll=”no” hundred_percent_height_center_content=”yes” equal_height_columns=”no” menu_anchor=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” status=”published” publish_date=”” class=”” id=”” link_color=”” link_hover_color=”” border_size=”” border_color=”” border_style=”solid” margin_top=”” margin_bottom=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” gradient_start_color=”” gradient_end_color=”” gradient_start_position=”0″ gradient_end_position=”100″ gradient_type=”linear” radial_direction=”center center” linear_angle=”180″ background_color=”” background_image=”” background_position=”center center” background_repeat=”no-repeat” fade=”no” background_parallax=”none” enable_mobile=”no” parallax_speed=”0.3″ background_blend_mode=”none” video_mp4=”” video_webm=”” video_ogv=”” video_url=”” video_aspect_ratio=”16:9″ video_loop=”yes” video_mute=”yes” video_preview_image=”” filter_hue=”0″ filter_saturation=”100″ filter_brightness=”100″ filter_contrast=”100″ filter_invert=”0″ filter_sepia=”0″ filter_opacity=”100″ filter_blur=”0″ filter_hue_hover=”0″ filter_saturation_hover=”100″ filter_brightness_hover=”100″ filter_contrast_hover=”100″ filter_invert_hover=”0″ filter_sepia_hover=”0″ filter_opacity_hover=”100″ filter_blur_hover=”0″][fusion_builder_row][fusion_builder_column type=”1_1″ layout=”1_1″ spacing=”” center_content=”no” link=”” target=”_self” min_height=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” hover_type=”none” border_size=”0″ border_color=”” border_style=”solid” border_position=”all” border_radius=”” box_shadow=”no” dimension_box_shadow=”” box_shadow_blur=”0″ box_shadow_spread=”0″ box_shadow_color=”” box_shadow_style=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” margin_top=”” margin_bottom=”” background_type=”single” gradient_start_color=”” gradient_end_color=”” gradient_start_position=”0″ gradient_end_position=”100″ gradient_type=”linear” radial_direction=”center center” linear_angle=”180″ background_color=”” background_image=”” background_image_id=”” background_position=”left top” background_repeat=”no-repeat” background_blend_mode=”none” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” filter_type=”regular” filter_hue=”0″ filter_saturation=”100″ filter_brightness=”100″ filter_contrast=”100″ filter_invert=”0″ filter_sepia=”0″ filter_opacity=”100″ filter_blur=”0″ filter_hue_hover=”0″ filter_saturation_hover=”100″ filter_brightness_hover=”100″ filter_contrast_hover=”100″ filter_invert_hover=”0″ filter_sepia_hover=”0″ filter_opacity_hover=”100″ filter_blur_hover=”0″ last=”no”][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
- Cassava Sciences Inc, can be described as another bio zombie with a “colourful” history, plodding along via toxic/dilutive financing.
- Some of the most notorious so-called “vulture” funds have been involved with the company over the last couple of years.
[/fusion_text][/fusion_builder_column][/fusion_builder_row][/fusion_builder_container][fusion_builder_container hundred_percent=”no” hundred_percent_height=”no” hundred_percent_height_scroll=”no” hundred_percent_height_center_content=”yes” equal_height_columns=”no” menu_anchor=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” status=”published” publish_date=”” class=”” id=”” background_color=”” background_image=”” background_position=”center center” background_repeat=”no-repeat” fade=”no” background_parallax=”none” enable_mobile=”no” parallax_speed=”0.3″ video_mp4=”” video_webm=”” video_ogv=”” video_url=”” video_aspect_ratio=”16:9″ video_loop=”yes” video_mute=”yes” video_preview_image=”” border_size=”” border_color=”” border_style=”solid” margin_top=”” margin_bottom=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” admin_toggled=”no”][fusion_builder_row][fusion_builder_column type=”1_2″ layout=”1_2″ spacing=”” center_content=”no” link=”” target=”_self” min_height=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” background_color=”” background_image=”” background_image_id=”” background_position=”left top” background_repeat=”no-repeat” hover_type=”none” border_size=”0″ border_color=”” border_style=”solid” border_position=”all” border_radius=”” box_shadow=”no” dimension_box_shadow=”” box_shadow_blur=”0″ box_shadow_spread=”0″ box_shadow_color=”” box_shadow_style=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” margin_top=”” margin_bottom=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” last=”no”][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Intro:
Since the end of 2019, Cassava Sciences Inc’s share price has experienced a considerable amount of volatility, nearly reaching a four year high in early October as well as this week. Like many small cap biotechs, SAVA (formerly known as PTIE) raises a few red flags that are worthwhile highlighting to anyone considering investing in the company. They include: a history of failure that stretches over two decades; a CEO that has pocketed millions of dollars in the process; and an abundance of highly questionable institutional investors.
[/fusion_text][/fusion_builder_column][fusion_builder_column type=”1_2″ layout=”1_2″ spacing=”” center_content=”no” link=”” target=”_self” min_height=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” background_color=”” background_image=”” background_image_id=”” background_position=”left top” background_repeat=”no-repeat” hover_type=”none” border_size=”0″ border_color=”” border_style=”solid” border_position=”all” border_radius=”” box_shadow=”no” dimension_box_shadow=”” box_shadow_blur=”0″ box_shadow_spread=”0″ box_shadow_color=”” box_shadow_style=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” margin_top=”” margin_bottom=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” last=”no”][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Security Details:
Symbol: SAVA
Current price:$10.69
Outstanding shares: 24.9 million
Market Cap: $266.2 million
[/fusion_text][/fusion_builder_column][/fusion_builder_row][/fusion_builder_container][fusion_builder_container hundred_percent=”no” hundred_percent_height=”no” hundred_percent_height_scroll=”no” hundred_percent_height_center_content=”yes” equal_height_columns=”no” menu_anchor=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” status=”published” publish_date=”” class=”” id=”” background_color=”” background_image=”” background_position=”center center” background_repeat=”no-repeat” fade=”no” background_parallax=”none” enable_mobile=”no” parallax_speed=”0.3″ video_mp4=”” video_webm=”” video_ogv=”” video_url=”” video_aspect_ratio=”16:9″ video_loop=”yes” video_mute=”yes” video_preview_image=”” border_size=”” border_color=”” border_style=”solid” margin_top=”” margin_bottom=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” admin_toggled=”no”][fusion_builder_row][fusion_builder_column type=”1_2″ layout=”1_2″ spacing=”” center_content=”no” link=”” target=”_self” min_height=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” background_color=”” background_image=”” background_image_id=”” background_position=”left top” background_repeat=”no-repeat” hover_type=”none” border_size=”0″ border_color=”” border_style=”solid” border_position=”all” border_radius=”” box_shadow=”no” dimension_box_shadow=”” box_shadow_blur=”0″ box_shadow_spread=”0″ box_shadow_color=”” box_shadow_style=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” margin_top=”” margin_bottom=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” last=”no”][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Company information:
Cassava Biosciences Inc. describes itself as “a clinical-stage drug development company. (its) expertise is to develop new product candidates and to guide these through various regulatory and development pathways in preparation for their potential commercialization”. Their lead drug candidate is Sumifilam (formerly known as PTI-125), an Alzheimer’s treatment that has been undergoing Phase 2 clinical trials over the last year or so, and press releases relating to said trails have coincided with notable increases in the stock’s volatility.
It is also worth noting that these trials have yielded far from positive results on first attempts, as the company’s most recent 10Q form states that the “Phase 2b study missed its pre-specified primary outcome, defined as a drug effect on cerebrospinal fluid (CSF) levels of tau protein and other biomarker assessments. The data set from that initial bioanalysis showed unnaturally high variability and other problems, such as no correlation among changes in levels of biomarkers over 28 days, even in the placebo group, and different biomarkers of disease moving in opposite directions in the same patient”. Yet the company has insisted on “conducting a comprehensive analysis of clinical results of our Phase 2b study, including evaluating the effects of PTI-125 on cognition” because the “data from the initial bioanalysis can be interpreted as anomalous and highly improbable”. Basically, the company’s CEO refused to accept the results and accused the lab of carrying out shoddy work. As expected the company conducted a second analysis of these Phase 2b trials carried out by an “academic” lab and it now shows “significant reductions ” in key biomarkers for Alzheimer’s disease, an announcement that is not in the least surprising considering the company’s long and turbulent history.
It should be pointed out that the company is no stranger to chronic failure. Before March 2019, Cassava Sciences Inc. was known as Pain Therapeutics (PTIE). This is an important, long and unsuccessful chapter in the company’s history and we will be taking a closer look at its latter years.
[/fusion_text][/fusion_builder_column][fusion_builder_column type=”1_2″ layout=”1_2″ spacing=”” center_content=”no” link=”” target=”_self” min_height=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” background_color=”” background_image=”” background_image_id=”” background_position=”left top” background_repeat=”no-repeat” hover_type=”none” border_size=”0″ border_color=”” border_style=”solid” border_position=”all” border_radius=”” box_shadow=”no” dimension_box_shadow=”” box_shadow_blur=”0″ box_shadow_spread=”0″ box_shadow_color=”” box_shadow_style=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” margin_top=”” margin_bottom=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” last=”no”][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Recent News:
[/fusion_text][/fusion_builder_column][/fusion_builder_row][/fusion_builder_container][fusion_builder_container hundred_percent=”no” hundred_percent_height=”no” hundred_percent_height_scroll=”no” hundred_percent_height_center_content=”yes” equal_height_columns=”no” menu_anchor=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” status=”published” publish_date=”” class=”” id=”” background_color=”” background_image=”” background_position=”center center” background_repeat=”no-repeat” fade=”no” background_parallax=”none” enable_mobile=”no” parallax_speed=”0.3″ video_mp4=”” video_webm=”” video_ogv=”” video_url=”” video_aspect_ratio=”16:9″ video_loop=”yes” video_mute=”yes” video_preview_image=”” border_size=”” border_color=”” border_style=”solid” margin_top=”” margin_bottom=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” admin_toggled=”no”][fusion_builder_row][fusion_builder_column type=”1_1″ layout=”1_1″ spacing=”” center_content=”no” link=”” target=”_self” min_height=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” background_color=”” background_image=”” background_image_id=”” background_position=”left top” background_repeat=”no-repeat” hover_type=”none” border_size=”0″ border_color=”” border_style=”solid” border_position=”all” border_radius=”” box_shadow=”no” dimension_box_shadow=”” box_shadow_blur=”0″ box_shadow_spread=”0″ box_shadow_color=”” box_shadow_style=”” padding_top=”” padding_right=”” padding_bottom=”” padding_left=”” margin_top=”” margin_bottom=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” last=”no”][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Financial lowlights:
Despite being a so-called “biozombie” (an unsuccessful biotech company that has been around for decades despite never getting a drug approved by the FDA), SAVA’s recent financial information is not all that atrocious. The company held over $25 million in cash and cash equivalents at the end of Q2 2020, a comfortable sum when one considers that the company lost a comparatively modest $1.1 million over the same time period. The company also has its debt under control as it amounts less than $1.5 million dollars. However, it is worth pointing out that this has come at a cost to many shareholders as the number of outstanding shares has increased from 21.8 million to nearly 25 million since the beginning of the year.
It could thus be said that dilution is perhaps the most concerning issue SAVA shareholders are facing, especially if one bears in mind the company filed a shelf registration for $200 million in March, which has been in effect since May. The notion that dilution is a cause of concern is further reinforced by the fact that “During the six months ended June 30, 2020, the Company received proceeds of $3.8 million from the exercise of 3.1 million warrants”. These warrants expire on February 17, 2021 for as low a strike price as $1.25 per share. As we will see, some of the institutional investors that hold warrants have rather worrying track records when it comes down to the share price performance of their investments over the long run.
[/fusion_text][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Before the root vegetable, there was Pain:
As previously mentioned, before changing its name to that of a root vegetable, Cassava Therapeutics went by the name of Pain Therapeutics (PTIE) and its lead drug candidate was Remoxy (an abuse deterrent for opioid addicts). Remoxy was in development for many years (nearly a decade and a half) and was subject to over 50 studies at a cost in excess of $100 million. Despite its clinical trials being “successful” the FDA rejected it four times. This infuriated CEO and founder Remi Barbier who went on to accuse the FDA of making “math errors, material mistakes or misrepresentations”.
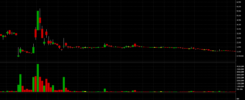
Around mid-2015 a class action lawsuit was filed against the company. The plaintiffs in the lawsuit claimed that “PTI(E) and the individual defendants caused shares in PTI(E) to trade at an artificially high price by concealing certain information from the public regarding the REMOXY FDA-approval process. Specifically, Plaintiff claims although Defendants knew they had failed to resolve problems with REMOXY’s stability that caused the FDA to reject the first REMOXY New Drug Application (NDA), Defendants led the public to believe those problems had been resolved while reapplying for FDA approval. Plaintiff further alleges that despite knowing the second NDA would also be rejected, PTI(E) rewarded the individual defendants with unjustifiable compensation packages it asked the shareholders to approve”. Basically, shareholders that had purchased overvalued shares were very “displeased” as they had been misled by statements made by the company’s management who in turn had been giving themselves hefty wages. The lawsuit was settled out of court for the sum of $8.5 million . As we can see, PTIE/SAVA has had a troubled “recent” history, but what about its overall history. We believe the share price chart since it became a publicly traded company nearly two decades ago speaks louder than words:
[/fusion_text][fusion_imageframe image_id=”12687|full” max_width=”” style_type=”” blur=”” stylecolor=”” hover_type=”none” bordersize=”” bordercolor=”” borderradius=”” align=”center” lightbox=”no” gallery_id=”” lightbox_image=”” lightbox_image_id=”” alt=”” link=”” linktarget=”_self” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” filter_hue=”0″ filter_saturation=”100″ filter_brightness=”100″ filter_contrast=”100″ filter_invert=”0″ filter_sepia=”0″ filter_opacity=”100″ filter_blur=”0″ filter_hue_hover=”0″ filter_saturation_hover=”100″ filter_brightness_hover=”100″ filter_contrast_hover=”100″ filter_invert_hover=”0″ filter_sepia_hover=”0″ filter_opacity_hover=”100″ filter_blur_hover=”0″]https://utopiacap.wordpress.com/wp-content/uploads/2024/07/f859a-sava-historic-share-price.png[/fusion_imageframe][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
It is also worth pointing out that Mr. Barbier has continued to benefit significantly from his position as SAVA/PTIE CEO. His current salary exceeds $900,000 and he currently owns 1,078,855 shares of company common stock a figure which is nearly half of what it was in February this year when it amounted to 1,924,247. It would thus appear that Mr. Barbier has likely benefited greatly from SAVA’s share price volatility this year. It could also indicate that the CEO himself does not have an awful lot of faith in the company’s future prospects, a claim that is further reinforced by the fact that in mid October 2020, he received $3 million in compensation despite Sumifilam’s unsuccessful clinical trials.
[/fusion_text][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Interesting “investors”:
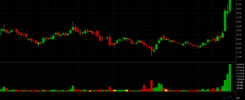
Unsurprisingly, Mr. Barbier is likely not the only individual or entity to have likely profited from the company’s recent share price volatility. A look at SAVA’s ownership filings will reveal some names speculative traders should be well familiar with. First up we have got Hudson Capital Management, arguably one of the most prolific so-called “vulture funds” currently operating in the realm of small caps due to the fact that the vast majority of companies in which it acquires a sizeable stake go on to exhibit highly disappointing share price performance over the long term, as well as having settled with the SEC for short selling regulations violations. This appears to be the exception with SAVA as the latest mention of Hudson Bay Capital in SAVA’s SEC filings dates back to February 2019 when it filed an ownership filing disclosing 1,176,471 shares of Common Stock issuable upon exercise of warrants. Since this date SAVA shares price has drastically increased.
[/fusion_text][fusion_imageframe image_id=”3396|full” max_width=”” style_type=”” blur=”” stylecolor=”” hover_type=”none” bordersize=”” bordercolor=”” borderradius=”” align=”center” lightbox=”no” gallery_id=”” lightbox_image=”” lightbox_image_id=”” alt=”” link=”” linktarget=”_self” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” filter_hue=”0″ filter_saturation=”100″ filter_brightness=”100″ filter_contrast=”100″ filter_invert=”0″ filter_sepia=”0″ filter_opacity=”100″ filter_blur=”0″ filter_hue_hover=”0″ filter_saturation_hover=”100″ filter_brightness_hover=”100″ filter_contrast_hover=”100″ filter_invert_hover=”0″ filter_sepia_hover=”0″ filter_opacity_hover=”100″ filter_blur_hover=”0″]https://utopiacap.wordpress.com/wp-content/uploads/2024/07/487e6-hudson_bay_sava_1.png[/fusion_imageframe][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Another “interesting” institutional investor that has been involved with the company over recent years is CVI Investments, an entity with a less than stellar track record when it comes to company’s in which it files SEC ownership filings.
Given that the company has issued millions of warrants to these institutional investors as well others similar ones such as Intracoastal Capital and Armistice Capital, we remain highly sceptical about the company’s financing partners and any positive impact their involvement could have on the share price.
[/fusion_text][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Red Flags:
[/fusion_text][fusion_imageframe image_id=”12690|full” max_width=”” style_type=”” blur=”” stylecolor=”” hover_type=”none” bordersize=”” bordercolor=”” borderradius=”” align=”none” lightbox=”no” gallery_id=”” lightbox_image=”” lightbox_image_id=”” alt=”” link=”” linktarget=”_self” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=”” filter_hue=”0″ filter_saturation=”100″ filter_brightness=”100″ filter_contrast=”100″ filter_invert=”0″ filter_sepia=”0″ filter_opacity=”100″ filter_blur=”0″ filter_hue_hover=”0″ filter_saturation_hover=”100″ filter_brightness_hover=”100″ filter_contrast_hover=”100″ filter_invert_hover=”0″ filter_sepia_hover=”0″ filter_opacity_hover=”100″ filter_blur_hover=”0″]https://utopiacap.wordpress.com/wp-content/uploads/2024/07/0cd00-sava-flags-2.png[/fusion_imageframe][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
Opinion:
SAVA is not a good investment. A history of persistent lack of success over two decades, questionable clinical trial data, settled class action lawsuits, regular dealings with so-called “vulture funds” and a CEO who earns close to $1 million a year (excluding share compensation) in spite of it all is the most concise way of describing this company. On November 7, the company will present the clinical data set from a Phase 2b study of Sumifilam. We expect this data set to continue the company’s long standing tradition of failure and questionable data, resulting in a share price decline next week.
[/fusion_text][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=”” animation_type=”” animation_direction=”left” animation_speed=”0.3″ animation_offset=””]
GET OUR NEWSLETTER DELIVERED TO YOUR INBOX:
[/fusion_text][fusion_text columns=”” column_min_width=”” column_spacing=”” rule_style=”default” rule_size=”” rule_color=”” hide_on_mobile=”small-visibility,medium-visibility,large-visibility” class=”” id=””][mailpoet_form id=”1″][/fusion_text][/fusion_builder_column][/fusion_builder_row][/fusion_builder_container]